ZyCoV-D® Overview
ZyCoV-D® is a DNA based vaccine for prevention of COVID-19. It comprises of a DNA plasmid vector carrying full length spike (S) gene region expressing SARS-CoV-2 spike (S) protein along with gene coding for signal peptide.1
The spike gene region was selected from submitted Wuhan Hu-1 isolate sequence (Genebank Accession No. MN908947.3).1
The S protein of the virus includes the receptor binding domain (RBD), responsible for binding to the human angiotensin converting enzyme-2 (ACE-2) receptor, which mediates the entry of virus inside the cell. The DNA plasmid construct was transformed into E. coli cells for large scale production.1
Therapeutic indication
ZyCoV-D® is indicated for active immunisation of individuals ≥12 years old for the prevention of coronavirus disease 2019 (COVID-19).1
Posology and method of administration
ZyCoV-D® vaccination schedule consists of 3 separate doses to be given at an interval of 28 days each (day 0, day 28 and day 56). Each dose consists of two shots of 0.1 ml each given by needle free injector (PharmaJet Tropis device) via intradermal route at two separate sites (preferably deltoid region of both the arms).1
Kindly refer to Medication Guide for Method of Administration
Contraindications
ZyCoV-D® is contraindicated in individuals known to have hypersensitivity to the active substance or to any of the excipients.1
Shelf-life
The expiry date of the vaccine is indicated on the label and packaging. Once opened, multi-dose vials should be used as soon as practically possible and within 6 hours when kept between +2ºC and +8ºC. All opened multidose vials of ZyCoV-D® should be discarded at the end of immunization session or within six hours whichever comes first.1
Storage and handling instructions
Store at 2° to 8°C. Multidose vials to be used within 6 hours of opening.1
How to Administer ZyCoV-D®
ZyCoV-D® has to be administered only by the Needle-Free Injector System (NFIS) using the PharmaJet Tropis device. It delivers the DNA vaccine into the cell via an intradermal route through a pressure device. The drug gets delivered between layers of the skin. Spring-powered injectors are used to deliver vaccine by means of a narrow stream of fluid that penetrates the skin in about 1/10 of a second.1,4
Vaccines administered by the Needle-Free Injection System (NFIS) are reported to be as immunogenic as when given with needle and syringe. Studies have shown that NFIS is as effective in delivering the vaccine, as traditional needle and syringes are across children, adolescents and adults.2-4
To learn more about ‘How to administer ZyCoV-D® you can click and download the Medication guide
Mechanism of Action of DNA Vaccines
The fundamental idea behind DNA vaccines is to induce immune responses against recombinant antigens encoded by genetically engineered DNA plasmids expressed in vivo.5 A plasmid is a small, circular, double-stranded DNA molecule that is distinct from a cell’s chromosomal DNA. The plasmid construct of ZyCoV-D® carries the spike-S gene of SARS-CoV-2. This pDNA enters host cells, where it remains in the nucleus as an episome; without getting integrated into the host cell DNA. The Plasmid DNA expresses the mRNA, which uses cell machinery to express spike antigen.6
Synthesis of SARS-CoV-2 spike (S) protein gene containing IgE signal peptide gene region and further cloning into pVAX-1 vector results in generation of SARS-CoV-2 DNA vaccine construct as shown below. 6


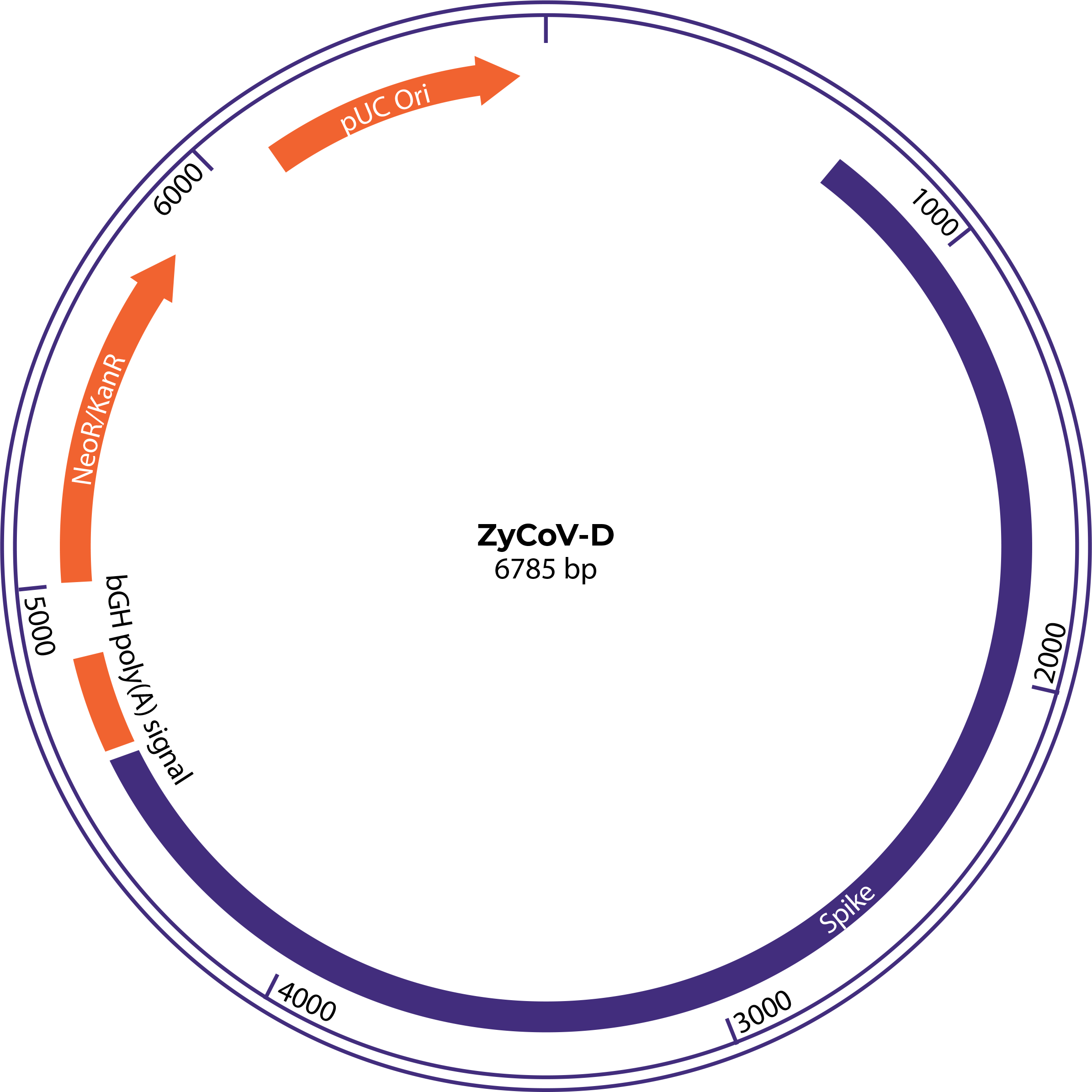
ZyCoV-D® synthetic DNA vaccine constructs. pVAX-1 vector containing SARS-CoV-2 spike gene insert 6
Mechanism of action of DNA vaccines is depicted below:6



carrying spike-S gene of interest enters host cells
Remains in the nucleus without getting integrated into host cell DNA
vaccine

protein is presented to APCs

Cross-priming and antigen presented to both CD4+ and CD8+ cells

Activated B cells produce antibodies (humoral response)

(cellular response)
Vaccines remain the key protective measure to achieve herd immunity to control the disease burden and stop COVID-19 pandemic. We have developed and assessed the immunogenicity and protective efficacy of ZyCoV-D® (a plasmid DNA based vaccine candidates) administered through Needle Free Injection System (NFIS) and syringe-needle (intradermal) with three dose vaccine regimens.7
A phase III, randomized, multi-centre, double blind, placebo controlled, study to evaluate efficacy, safety and immunogenicity of Novel Corona Virus -2019-nCov vaccine candidate of M/s Cadila Healthcare Limited
ZyCoV-D® is a DNA vaccine candidate, which comprises a plasmid DNA carrying spike-S gene of SARS-CoV-2 virus along with gene coding for signal peptide. The spike(S) region includes the receptor-binding domain (RBD), which binds to the human angiotensin converting Enzyme (ACE)-2 receptor and mediates the entry of virus inside the cell.8
This study has been conducted in India on 28216 healthy volunteers.
They were randomized in 1:1 ratio (14108 in test arm and 14108 in placebo arm).8
Phase III | |
N | 28216 (1:1 Vaccine & Placebo) |
Age | 12+ years 1405 subjects: 12 – 17 years 3000 subjects: >60 / comorbidities |
Number of Doses | Three (Day 0, Day 28 and Day 56) |
Dose | 2 mg with Pharmajet (0.1ml ID in either deltoid region) |
Safety and tolerability | Very well tolerated |
SAE/Death | No vaccine related SAE/death |
Efficacy Evaluation (Interim)8
Overall Efficacy after 84 days (Interim analysis of 81 cases as per protocol) | 66.6 % |
Efficacy for MILD cases after 84 days (based on 78 mild cases) | 64.9% |
Efficacy for MODERATE cases after 84 days | 100% |
Efficacy for SEVERE cases (after 2nd dose) | 100% |
Immunogenicity Evaluation8
ZyCoV-D® | Placebo | |
Seroconversion rate IgG titer | 93.33% at Day 84 | 52.31% at Day 84 |
IgG titer (EU) (GMT, 95% CI) | 952.67 (707.9, 1282.0) | 154.82 (91.25, 262.7) |
IgG titer (GMFR, 95% CI) | 136.09 (101.11, 183.1) | 22.12 (13.04, 37.53) |
Cell Mediate Immune Response8
- IFN-γ levels measured by ELISpot shows almost 13-15 fold increase post vaccination in the vaccine arm
The overall efficacy of the vaccine in interim analysis on day 84 after the last dose was found to be 66.6% along with 100% efficacy against moderate and severe cases. IFN-γ levels measured by ELISpot shows almost 13-15 fold increase post vaccination in the vaccine arm.8
- DNA based vaccine is one of the most preferred amongst new generation vaccines. ZyCoV-D® used a plasmid vector which was constructed to be consistent with the US-FDA document, The vector has also been used in numerous other clinical trials globally for other vaccines in past and found to be safe.
- Current nonclinical and clinical databases support that prior concerns about integration, autoimmunity and immunopathology have not been borne out.
- It is now accepted that DNA vaccines to date do not persist or even bio-distribute throughout the body of the vaccine recipient when delivered parenterally into muscle, subcutaneous tissue or various dermal layers.9 Biodistribution studies are waived for DNA vaccines produced by inserting a novel gene into a plasmid vector.10
- In comparison to the published clinical data of other vaccines, ZyCoV-D® has a better safety profile in terms of systemic and local adverse events.6,11,12
- For ZyCoV-D® administration we use PharmaJetTropis device for delivery of the vaccine, which is a spring powered injector delivering vaccine by means of a narrow stream of fluid that penetrates the skin in about 1/10th of a second making it faster and safer.2-4
- Comparative Safety Profile of ZyCoV-D® Vaccine as compared to other vaccines as per the published systematic review and meta-analysis13
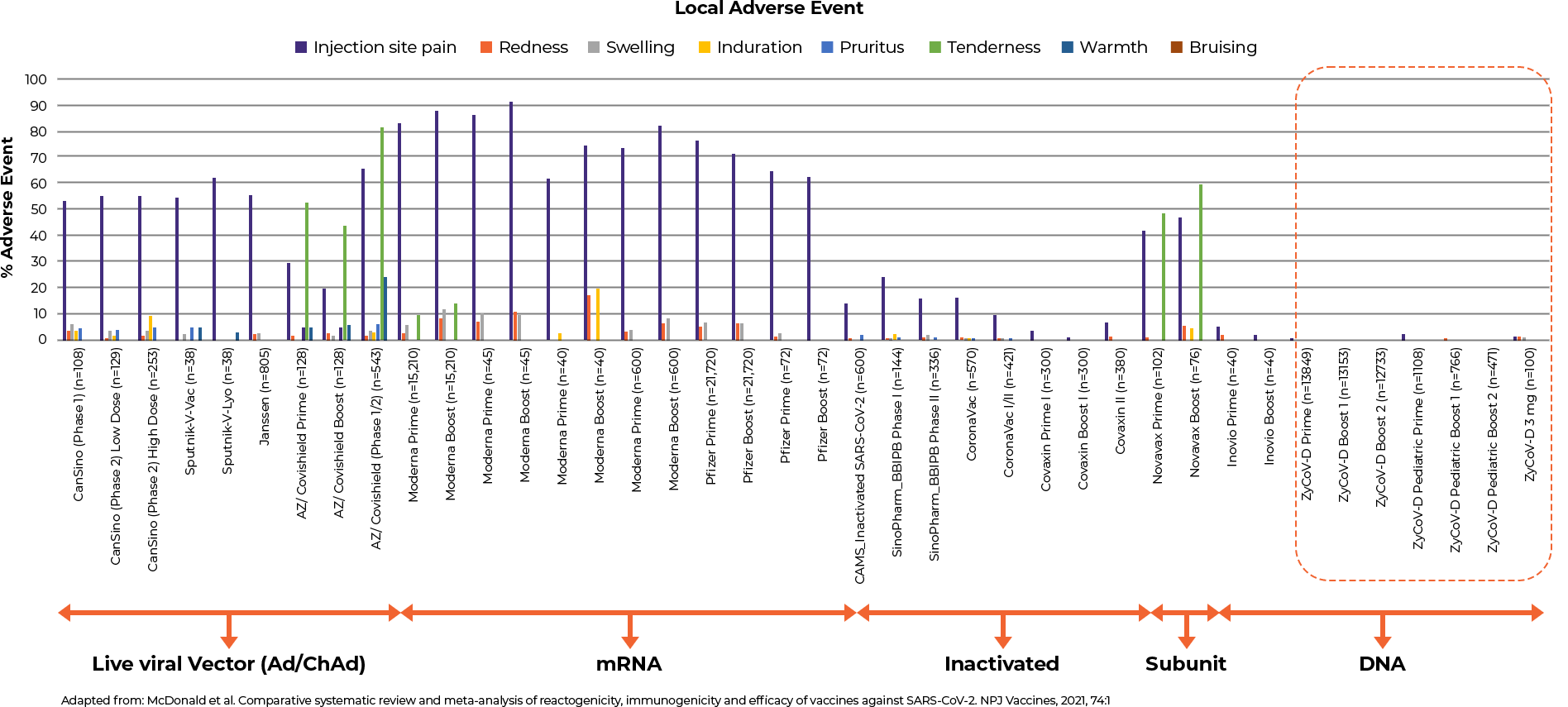
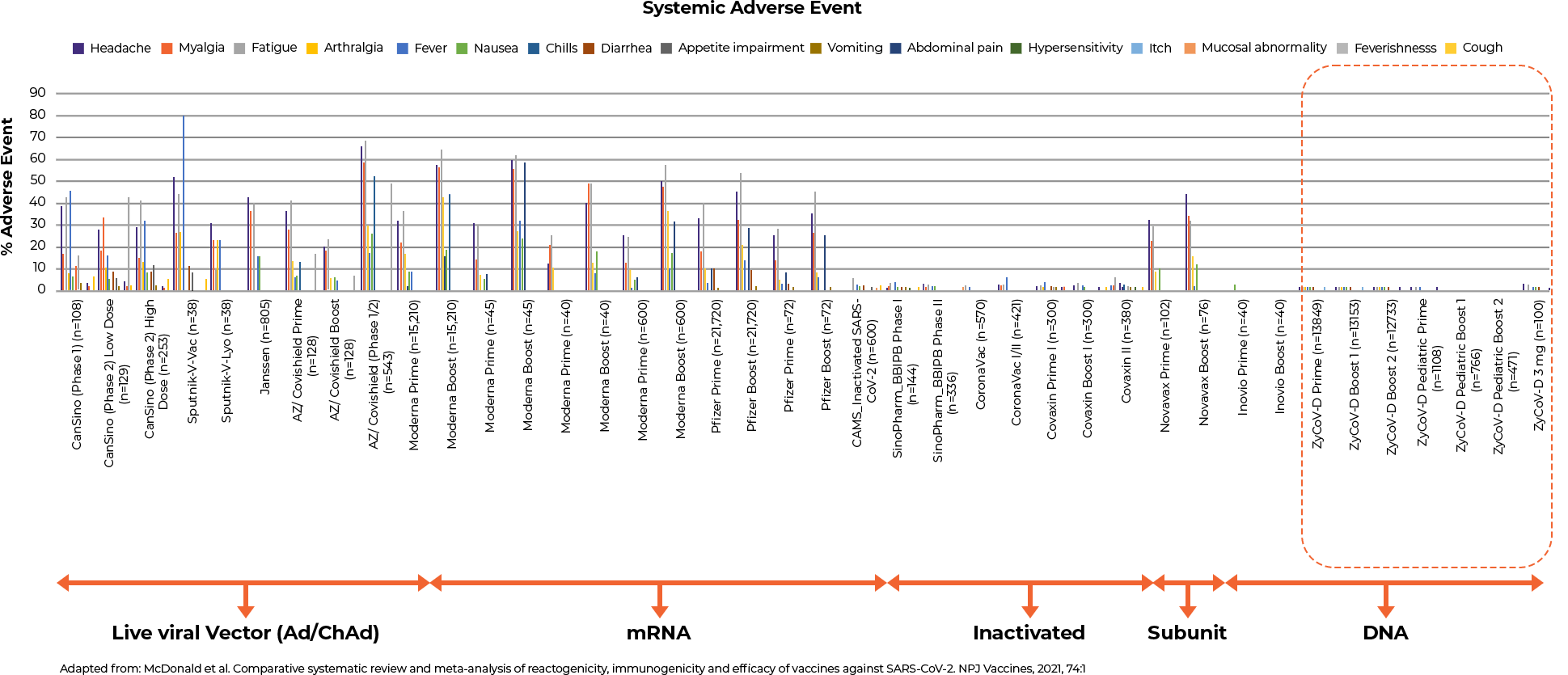
- ZyCoV-D® has a better safety profile compared to the published data of other vaccines in terms of systemic and local adverse events
The emergence of new strains of virus has raised the doubts about efficacy of vaccines which were already approved for emergency use authorization.11
DNA vaccines are based on plug and play platform, which allows rapid development of new constructs in case mutant strains develop, and possibility of generating a new vaccine candidate in very short time, thus providing protection against mutated viral strains.11
Safety and Immunogenicity of a DNA SARS-CoV-2 vaccine (ZyCoV-D®): Results of an open-label, non-randomized phase I part of phase I/II clinical study by intradermal route in healthy subjects in India
This first-in-human trial showed that the DNA vaccine was tolerable and immunogenic in healthy adults. The DNA vaccine candidate induces antibody response against SARS-CoV-2 spike (S) protein, following immunization with three doses administered 28 days apart.11
This study has also evaluated safety and immunogenicity of DNA vaccine administered by two different methods of administration.11
This was a single-center, open-label, non-randomized, Phase 1 trial in India between July 2020 and October 2020.
Healthy adults aged between 18 and 55 years were sequentially enrolled and allocated to one of four treatment arms in a dose escalation manner.11
Three doses of vaccine were administered 28 days apart and each subject was followed up for 28 days post third dose to evaluate safety and immunogenicity.11
Out of 126 individuals screened for eligibility, 48 subjects (mean age 34.9 years) were enrolled and vaccinated in the Phase 1 study. Overall, 12/48 (25%) subjects reported at least one AE (i.e. combined solicited and unsolicited) during the study.11
There were no deaths or serious adverse events reported in Phase 1 of the study.11
The proportion of subjects who seroconverted based on IgG titers on day 84 was 4/11 (36.36%), 4/12 (33.33%), 10/10 (100.00%) and 8/10 (80.00%) in the treatment Arm 1 (1 mg: Needle), Arm 2 (1 mg: NFIS), Arm 3 (2 mg: Needle) and Arm 4 (2 mg: NFIS), respectively.11




Figure 1: Neutralizing antibody titres with error bars, representing geometric mean (95%CI) at baseline (Day 0) and Day 84 with comparative Nab of convalescent serum.11
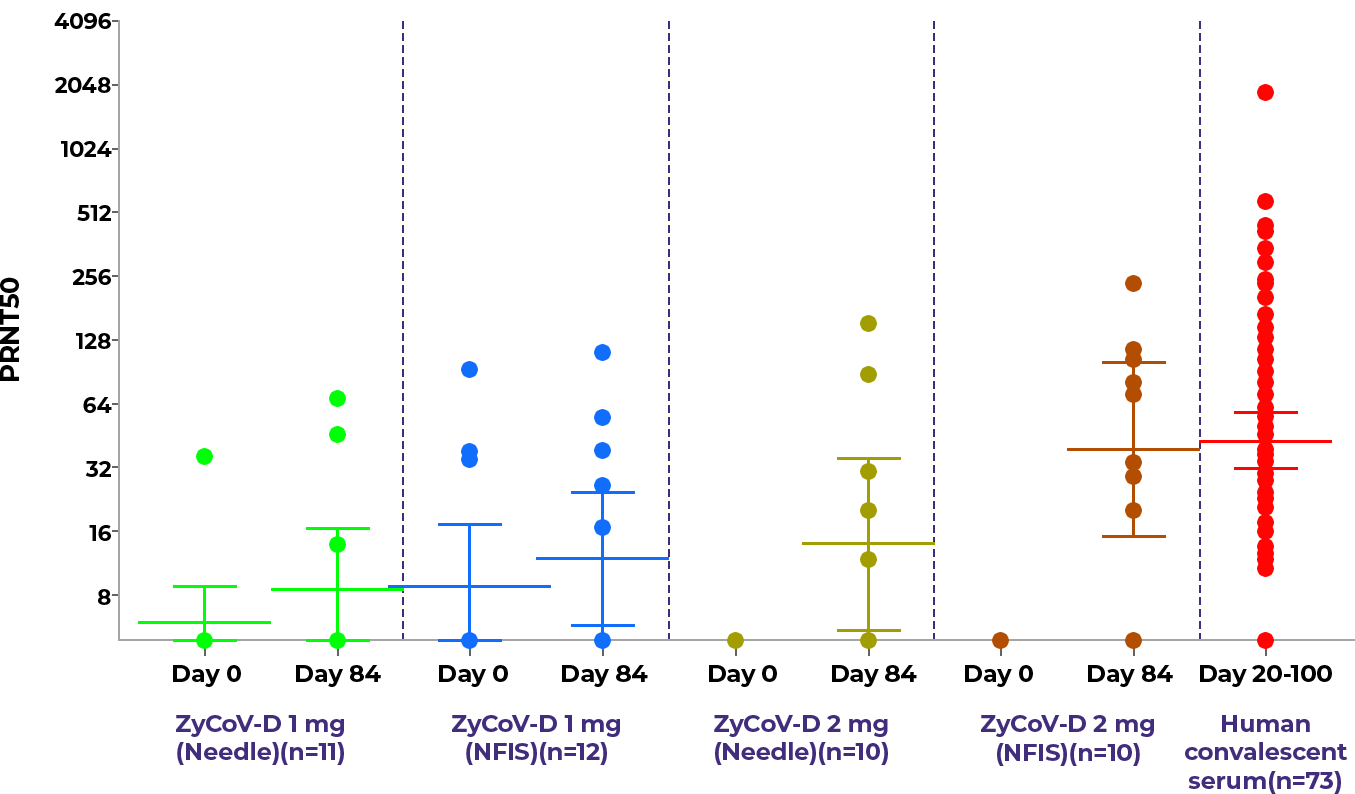
ZyCoV-D® vaccine is found to be safe, well-tolerated and immunogenic in the Phase 1 trial.11
Phase I 11 | Phase II 11 | |
N | 48 | 1000 |
Age | 18-45 years | 12+ years |
Number of Doses | Three (Day 0, Day 28 and Day 56) | Three (Day 0, Day 28 and Day 56) |
Highest dose tested ID (with needle / syringe & Pharmajet) | 2 mg | 2 mg |
Safety and tolerability | Very well | Very well |
SAE/Death | No SAE/death | No vaccine related SAE/No death |
Seroconversion rate IgG titer (2 mg Pharmajet) | 80% at Day 84 80% at Day 210 | 91.28% at Day 84 88.34% at Day 224 |
Seroconversion rate NAB titer (2 mg Pharmajet) | 80% at Day 84 80% at Day 210 (5.5 months after last dose) | 88.2% at Day 84 83.35% at Day 224 (06 months after last dose) |
NAB titer (2 mg Pharmajet) | 39.17 at Day 84 64.76 at Day 210 | 126.00 at Day 84 92.18 at Day 224 |
Majority of SARS-CoV-2 vaccine studies describe the protection conferred by the vaccination before waning of the acute phase immune response. The long-term immunological memory induced by these vaccine candidates yet needs to be explored as studied in case of few candidates. This is important as SARS-CoV-2 can cause asymptomatic or mild disease in humans without inducing significant antibody response.6
Immunogenicity and protective efficacy of ZyCoV-D® vaccine candidates in rhesus macaques against SARS-CoV-2 infection
The current study, evaluated the immunogenicity of ZyCoV-D® DNA vaccine candidates in rhesus macaques and the protective efficacy against SARS-CoV-2 infection post 14 weeks of first immunization dose.3
The vaccine candidate 2 mg dose administered using Needle Free Injection System (NFIS) elicited a significant immune response with development of SARS-CoV-2 S1 spike region specific IgG and neutralizing antibody (NAb) titers during the immunization phase and significant enhancement in the levels after the virus challenge.7
In 2 mg NFIS group the IgG and NAb titers were maintained and showed gradual rise during the immunization period (15 weeks) and till 2 weeks after the virus challenge.7
It also conferred better protection to macaques evident by the viral clearance from nasal swab, throat swab and bronchoalveolar lavage fluid specimens in comparison with macaques from other immunized groups.7
In contrast, the animals from placebo group developed high levels of viremia and lung disease following the virus challenge. Besides this, the vaccine candidate also induced increase lymphocyte proliferation and cytokines response (IL-6, IL-5).7
The administration of the vaccine candidate with NFIS generated a better immunogenicity response in comparison to syringe-needle (intradermal route). The study demonstrated immunogenicity and protective efficacy of the vaccine candidate, ZyCoV-D in rhesus macaques.7
DNA vaccines are attractive because they ensure appropriate folding of the polypeptide, produce the antigen over long periods, and do not require adjuvants. These host-synthesized antigens then can become the subject of immune surveillance in the context of both major histocompatibility complex class I (MHC I) and MHC II proteins of the vaccinated individual.6
Immunogenic potential of DNA vaccine candidate, ZyCoV-D® against
SARS-CoV-2 in animal models
Based on the animal studies proof-of-concept has been established and preclinical toxicology (PCT) studies were conducted in rat and rabbit model.6
The candidate vaccine ZyCoV-D® comprised of a DNA plasmid vector carrying the gene encoding the spike protein (S) of the SARS-CoV-2 virus. The S protein of the virus includes the receptor binding domain (RBD), responsible for binding to the human angiotensin converting enzyme (ACE-2) receptor. The DNA plasmid construct was transformed into E. coli cells for large scale production.6
The immunogenicity potential of the plasmid DNA has been evaluated in mice, guinea pig, and rabbit models by intradermal route at 25, 100 and 500 µg dose.6
DNA vaccine candidate generates robust response and neutralizes the SARS CoV-2 virus conferring protective immunity against infection.6
The usefulness and efficiency of a spring-powered, needle-free Injection System (NFIS) for delivering ZyCoV-D® vaccine in rabbits was also demonstrated in the study.6
Preliminary animal study demonstrates that the candidate DNA vaccine induces antibody response including neutralizing antibodies against SARSCoV-2 and also provided Th-1 response as evidenced by elevated IFN-γ levels.6
Key Developments6,7,11,12





fast-track discovery
efforts using in-house
DNA platform

developed


published

for >12 years
and above in India
initiated
trial completed
clinical recruitment
completed


1/2 study in India

demography
12 years & older
& well tolerated with no
Grade 3/4 AE/SAE
neutralized obtained
clinical trial in India
Volunteers
Volunteers
Evolution of DNA vaccines14
DNA vaccine encoding HIV-1 clade B gag, pol, env (PENNVAX-B) with/without Molecular IL – 12 is safe and immunogenic. EP and pIL- 12 is dose – sparing. First demonstration that DNA +EP induces responses as potent as viral vectored vaccines. HVTN 070/080
HPV E6/E7 DNA vaccines is safe and efficacious against cervical neoplasia in human patients
VEEV DNA vaccine is safe, immunogenic and well tolerated in human patients
DNA vaccine encoding HIV-1 clad B gag, pol, env (PENNVAX-B) with Molecular IL – 12 induced almost 100% vaccine response rate with durable responses. ID- EP is as potent as IM-EP with 1/5th dose. HVTN 098 (ClinicalTrials.gov Identifier: NCTO2431767) Wild-type and JEV-chimeric ZIKV jet delivered DNA vaccines are safe and well tolerated in adult volunteers VRC319.320
First- in – human ZIKV DNA vaccine is safe and immunogenic in humans 6.5 months bench-to-clinic. Influenza HA DNA prime via Biojector jet delivery followed by trivalent inactivated influenza boost (IIV3) in adults and children aged 6-17 years. First US study of a DNA vaccine in children. VRC 702 DNA vaccine+ pembrolizumab promotes anti-tumor responses in prostate cancer patients
Influenza HA DNA prime via Biojector jet delivery followed by trivalent inactivated influenza boost (IIV3) healthy adults. VRC 703
Intradermal EP of SynCone EBOV vaccine promotes cellular and humoral response in humans. 18 months beach-to-clinic
MERS-CoV DNA vaccine phase I trial vaccine is safe and immunogenic
Anti- ZIKV DMAb clinical trial begins February 2019 (clinicaltrials. Gov identifier :NCTO3831503)
Latest DNA vaccine against coronavirus disease 2019 (COVID-19). Intradermal DNA vaccine submitted for DCGI approval.
World’s first plasmid DNA based vaccine for human use, against COVID-19 (pDNA vaccine) going to be approved in India.
Differences Between Plasmid DNA Vaccine and Other Vaccine platforms
DNA vaccines

mRNA vaccines

Protein subunit

Viral vectors

Live attenuated virus

Inactivated virus

Virus like particles (VLPs)

Advantages of Intradermal Needle Free Delivery1,2-4,21,22










